Best calcium for chickens: Seashells, limestone or oysters?
The in vitro solubility of Morgan Agro sea shells in relation with the egg shell quality of laying hens.
Introduction
Laying hens need on daily basis a large amount of calcium (Ca) in the feed, mainly due to the formation of the egg shell. For one egg, approximately 2 – 2,5 grams calcium is needed. The total amount of calcium in the blood is only about 25 mg. This means that during the 16 – 20 hours egg formation, there needs to be a constant supply of calcium to the blood, to be able to form the egg shell. The calcium in blood originates from the digestion of calcium ingredients in the small intestine and the reserves in the bone. During the night time, when the major part of the egg shell is formed, the hen doesn’t consume any feed. During the night time, the major part of the calcium will therefore be mobilised from the bones and to a lesser extent from the available calcium ingredients in the intestine. When hens grow older, the efficiency of mobilisation of calcium from bones decreases. This means that older hens can show a decreased egg shell quality, because they face problems with the calcium supply to the blood during the egg shell formation in the night time. The older hens must therefore depend for a larger extend on the digestible calcium ingredients in the intestine.
The importance of solubility of calcium sources
It seems therefore important to use a calcium ingredient in the feed, from which the calcium will become available gradually during time in the intestinal tract, also during the night time. Through this, the calcium level in the blood during night time will originate for a larger extent from the calcium digestion in the intestine and to a lesser degree from bone mobilisation. The hen will be less dependent on the calcium mobilisation from the bone.
In the following table the pH values and retention time of feed in several intestinal parts are given.
| GIT section | Retention time | pH |
|---|---|---|
| Crop | 30 | 5.5 |
| Gizzard | 90 | 2.5-3.5 |
| Duodenum (first part of small intestine | 5-8 | 5-6 |
| Jejunum (middle part of small intestine | 20-30 | 6.5-7.0 |
As shown in the table above, the retention time of feed and calcium supplements in the gizzard is quite long. Research (Zhang & Coon, 1997) showed that larger limestone particles have a lower in vitro solubility and are retained in the gizzard for a longer time. Consequently, the longer retention time in the gizzard resulted in a higher availability of calcium for the laying hen.
Zhang and Coon (1997) studied the effect of dietary calcium level, particle size and in vitro limestone solubility on in vivo limestone solubilization and limestone retention in the gizzard of laying hens. The retention of calcium in the gizzard of laying hens is mainly influenced by the particle size and porosity of the calcium source. The study showed that calcium ingredients with a low in vitro digestibility have a higher retention time in the gizzard, leading to an increased in vivo solubility. Therefore, it was suggested that a prolonged retention (of large particle) calcium supplements in the gizzard makes calcium more available during the period of shell formation throughout the night. Similarly, research of Rao & Roland (1990) showed a higher availability of calcium in the small intestine when limestone with larger particles was used.
To obtain an indication of the availability of calcium from several calcium ingredients in the intestine of laying hens, an in vitro test was performed with several calcium ingredients. We conducted two experiments, as given below.
In vitro experiments
In two in vitro experiments carried out in The Netherlands, we studied the solubility of Morgan Agro seashells, oyster shells, finely ground limestone and coarse limestone.
Experiment 1
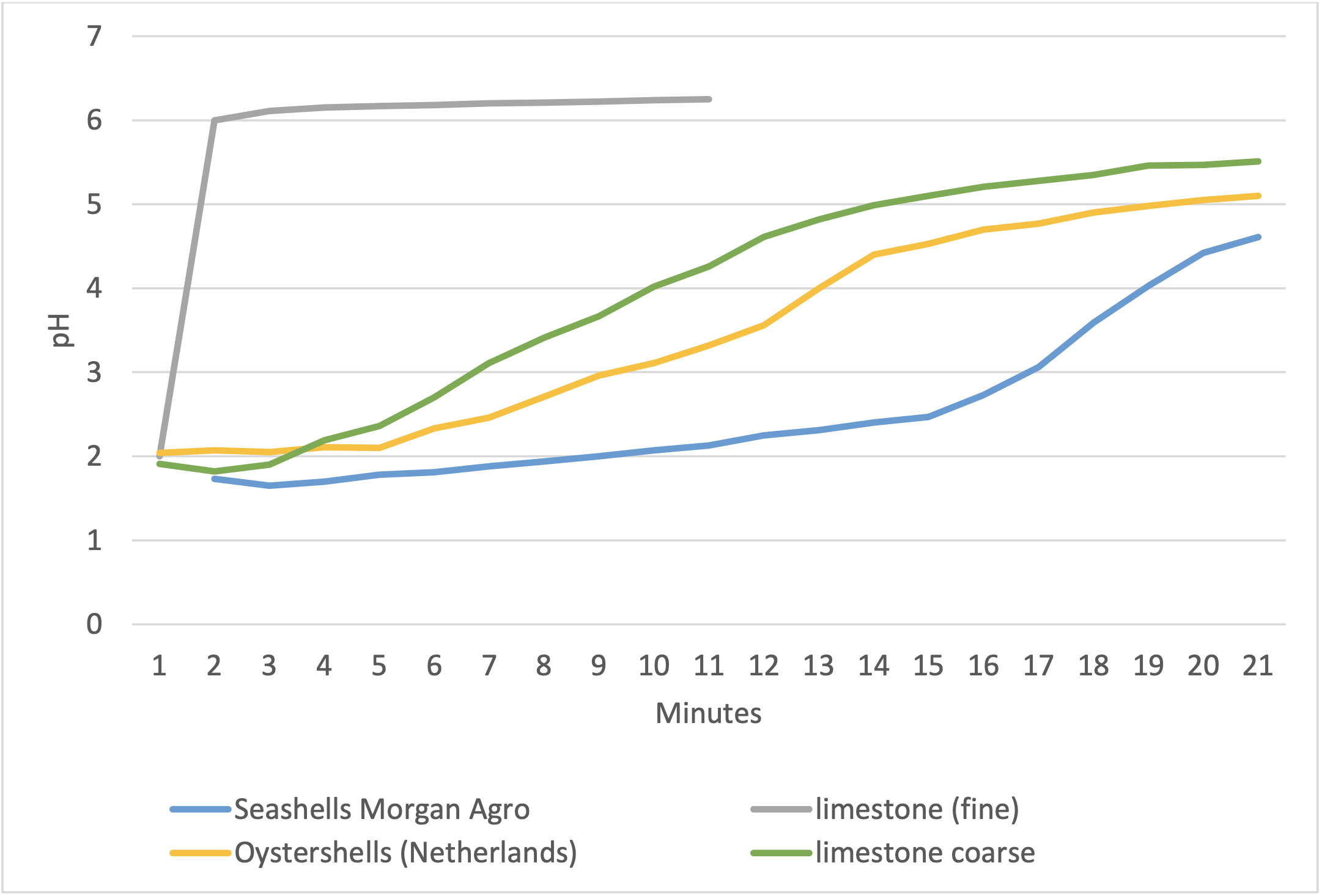
5 grams of the several calcium ingredients were added to 150 grams HCl solution. The pH values were recorded for a period of 20 minutes, as shown in the figure below.
The Morgan Agro seashells remained stable during a longer time period compared to the coarse limestone and oystershells. The fine limestone reacted immediately, followed by a sharp increase in pH value. This indicates a more gradual solubility in an acidic environment of the Morgan Agro seashells compared to limestone and oystershells.
Experiment 2
Calcium carbonate has a poor solubility in water. In, for instance, the gizzard of laying hens, the calcium will become soluble because of the available HCl, with the following reaction:
CaCO3 + 2 HCl→CaCl2 + H2O + CO2
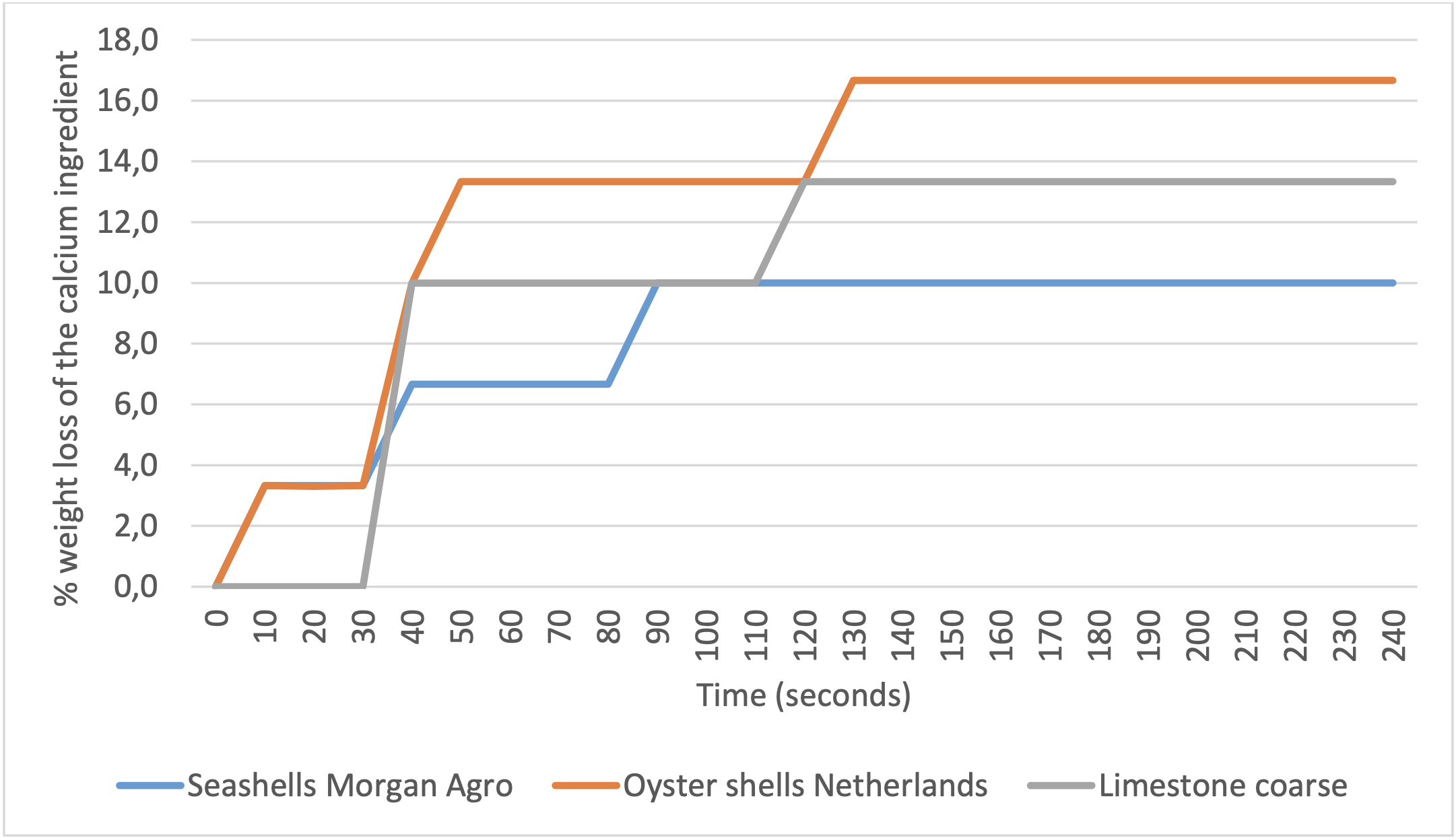
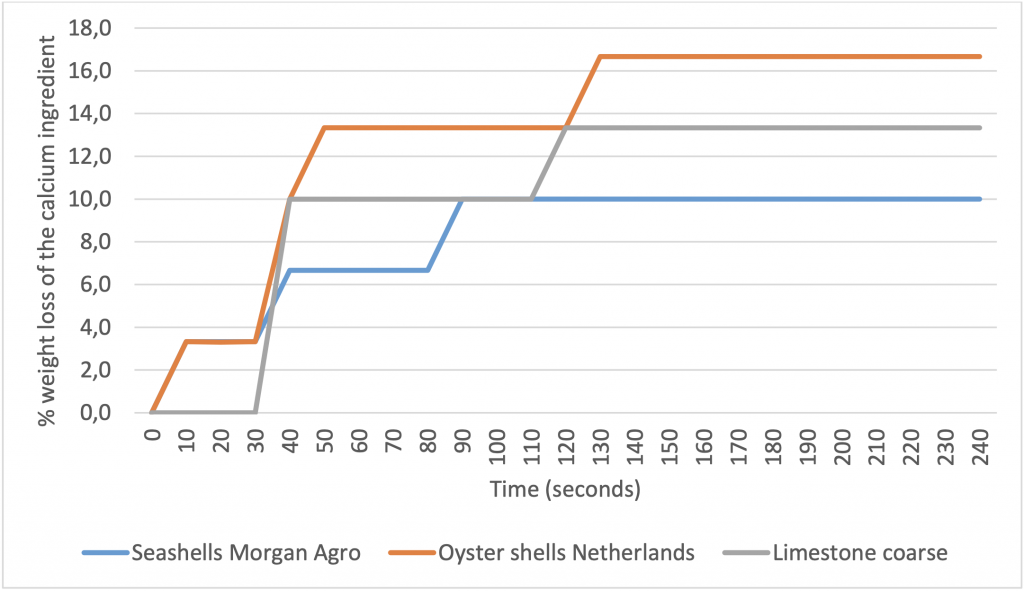
3 grams of the several calcium ingredients were added to 25 ml HCl solution (pH 0.69); during this time, the formed CO2 is lost and will lead to a reduction in weight. In the following graph, the % weight loss of the 3 gram calcium ingredients is visualised during a period of 4 minutes (the fine limestone is not shown in this graph because of the very strong reaction and rapid loss of CO2 at the start of the reaction).
Like experiment 1, in this experiment the Morgan Agro seashells showed a more gradual solubility in an acidic environment compared to oystershells and coarse limestone particles.
Conclusions
- Our in vitro work shows that Morgan Agro seashells have a more gradual solubility in an acidic environment compared to fine and coarse limestone and oystershells.
- It can be hypothesized that the more gradual solubilisation of the Morgan Agro seashells will lead to a longer retention time in the gizzard, and a better egg shell quality due to a more stable calcium supply in the small intestine.
- The major part of the laying hen egg shell is formed during the night time when no feed is consumed.
- The calcium level in the blood of laying hens can be regulated via the available calcium in the intestine and mobilisation from the bones.
- During night time, a larger part of the needed calcium for egg shell formation is obtained from bone mobilisation, compared to day time.
- When hens grow older, the efficiency of mobilisation of calcium from bones decreases, which can lead to poor egg shell quality.
- It is recommended (especially for older hens) to use a calcium ingredient from which the calcium will become available gradually during the time in the intestinal tract. The hen will be less dependent on the calcium mobilisation from the bone.
- Calcium ingredients with a poor solubility and large particle size have an increased retention time in the gizzard and ultimately a higher calcium availability for the animal.
Where to buy seashells?
Morgan Agro provide the highest quality seashells from the Caspian Sea. Available in retail packs of 2 KG and 15 KG, as well as bulk packing of 1 MT. The product is a great organic calcium supplement for chickens, are a natural source with the appropriate particle size, and are a real solution for the problem of weak egg shells in laying chickens. Click here to contact us.
Sources:
- Rao, KS and DA Roland, 1990. In vivo limestone solubilization in commercial Leghorns: role of dietary
calcium level, limestone particle size, in vitro limestone solubility rate, and the calcium status of the hen. Poultry Science 69: 2107-6. - Zhang, B. and C. Noon, 1997. The relationship of calcium intake, source, size, solubility in vitro and in vivo and gizzard limestone retention in laying hens. Poultry science 76: 1702-1706